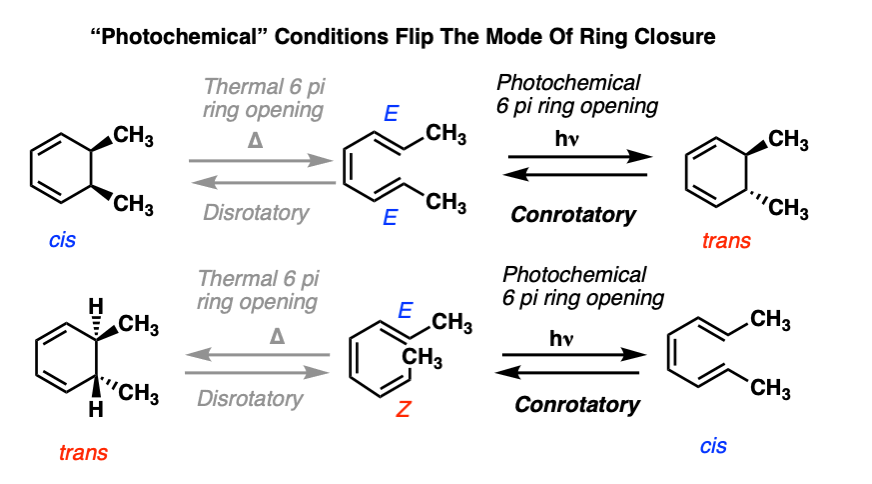
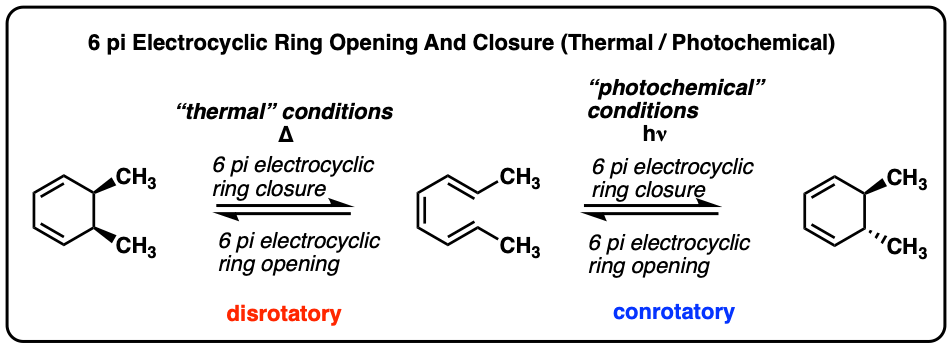
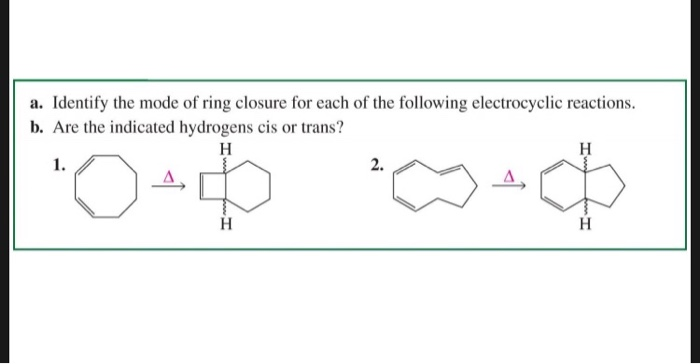
FeCl3‐Catalyzed Ring‐Closing Carbonyl–Olefin Metathesis - Ma - 2016 - Angewandte Chemie International Edition - Wiley Online Library
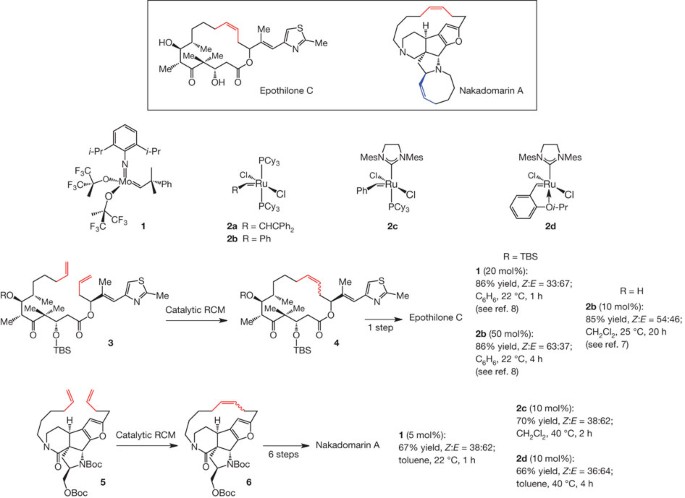
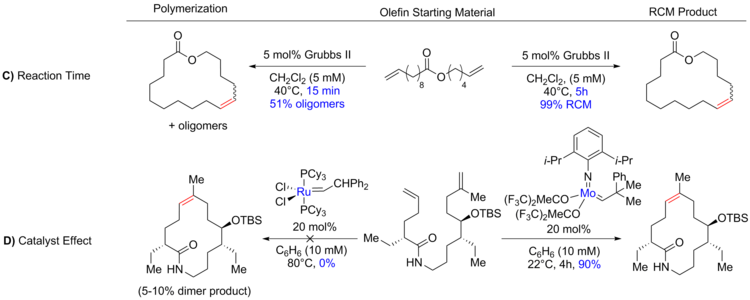
Synthesis of macrocyclic natural products by catalyst-controlled stereoselective ring-closing metathesis | Nature
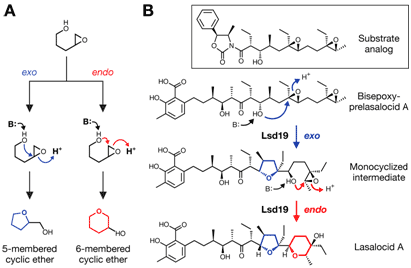
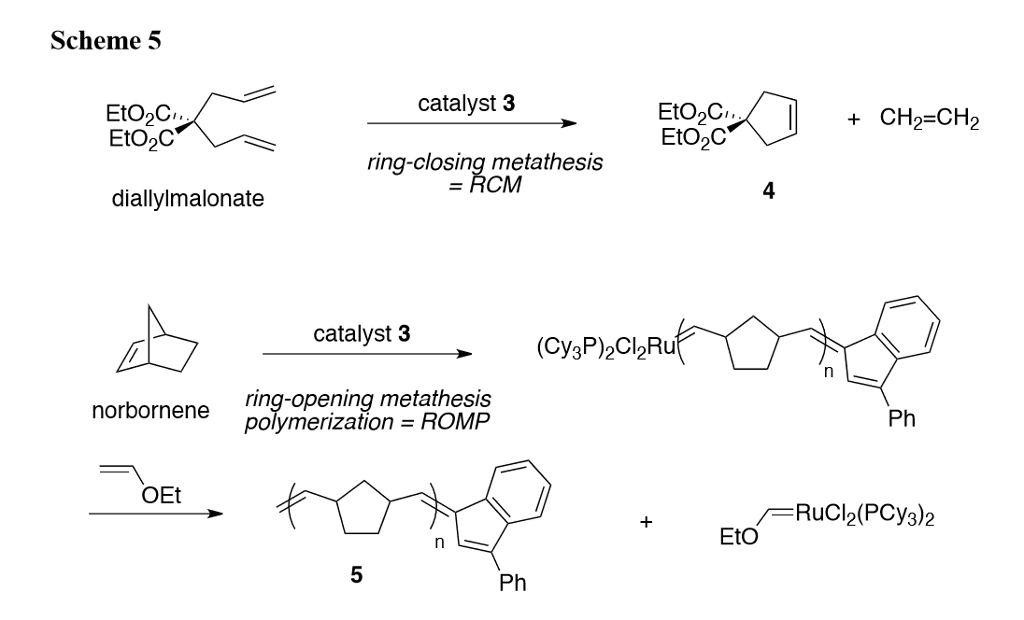
Structure of an Enzyme Catalyzing an Unfavorable Ring Closure Revealed | Stanford Synchrotron Radiation Lightsource