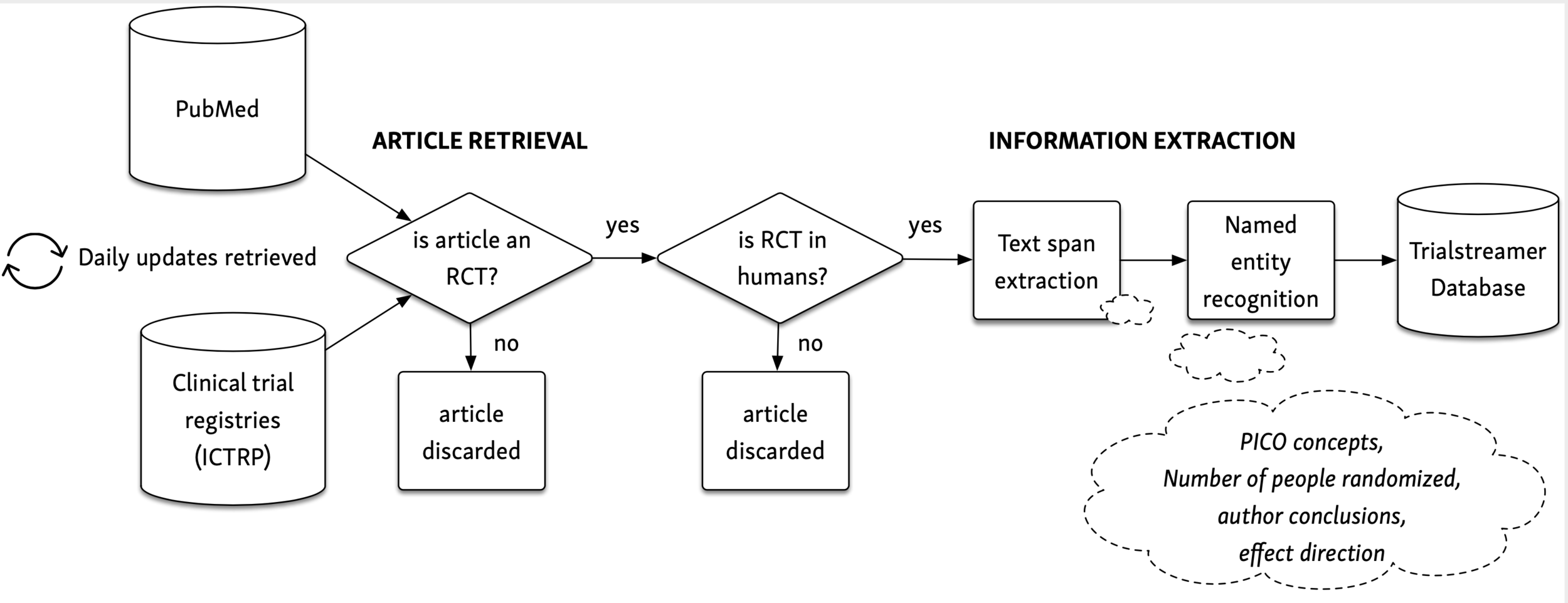
Generalizability of findings from randomized controlled trials is limited in the leading general medical journals - Journal of Clinical Epidemiology
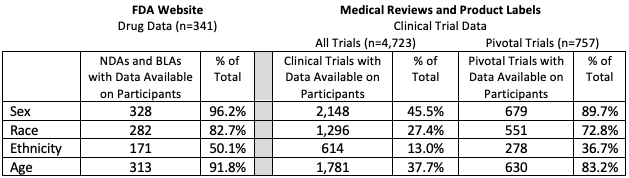
Demographic Disparities in Patient Samples for Drugs and Biologics Approved by FDA Between 2007-2017

Evidence-informed recommendations to reduce dissemination bias in clinical research: conclusions from the OPEN (Overcome failure to Publish nEgative fiNdings) project based on an international consensus meeting – topic of research paper in

Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial - The Lancet Infectious Diseases

Ongoing Clinical Trials for the Management of the COVID-19 Pandemic: Trends in Pharmacological Sciences

Accessibility of trial reports for drugs stalling in development: a systematic assessment of registered trials – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on

PDF) Clinical Trial Evidence Supporting US Food and Drug Administration Approval of Novel Cancer Therapies Between 2000 and 2016
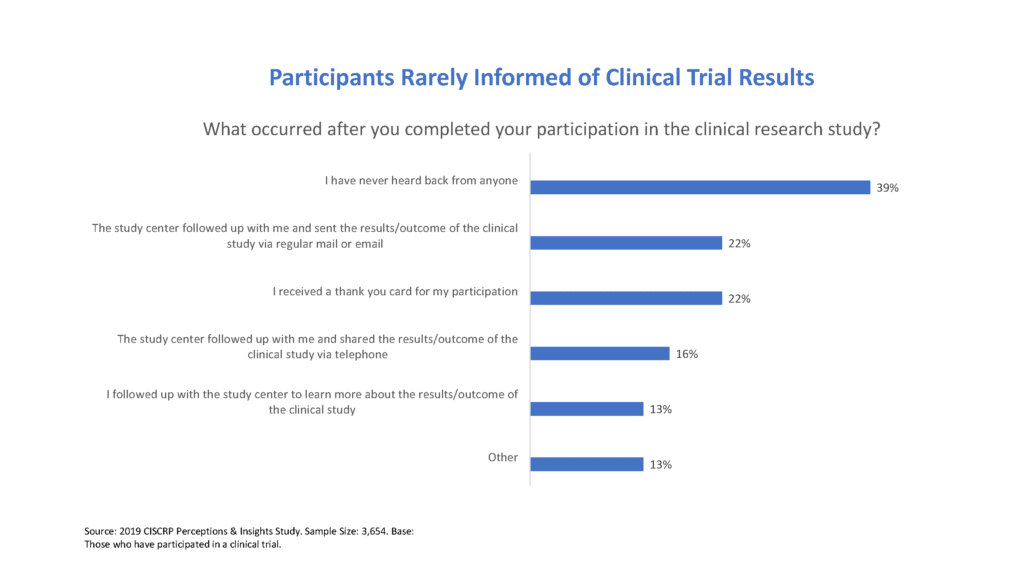
CISCRP's 2019 Perceptions and Insights Study - Center for Information & Study on Clinical Research Participation