
Teva Pharmaceutical | $TEVA Stock | Shares Rally After Winning FDA Approval for Migraine Therapy - Warrior Trading News

Frontiers | Pooled Analysis on the Effectiveness and Safety of Lipegfilgrastim in Patients With Urological Malignancies in the Real-World Setting | Oncology
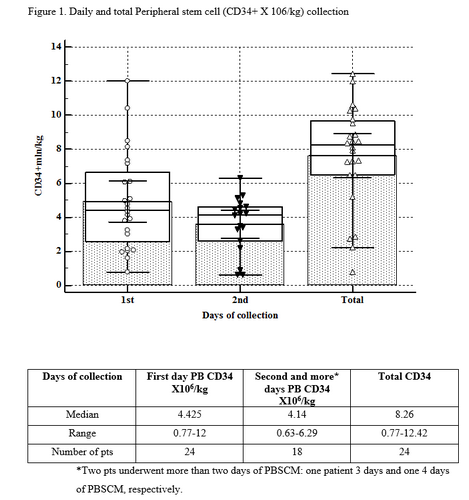
LONG-ACTING G-CSF (LIPEGFILGRASTIM, “LONQUEX”) FOR STEM CELL.... EHA Library. Danylesko I. Jun 15 2019; 267164

LONQUEX Trademark of TEVA Pharmaceutical Industries Ltd. Serial Number: 85427862 :: Trademarkia Trademarks
.png)
G-CSF (Granulocyte Colony Stimulating Factors) Market Analysis, Size And Trends Global Forecast To 2022-2030

Immunogenicity assessment of PEGylated proteins, Lonquex, a PEGylated G-CSF case study - ScienceDirect

Fillable Online newworldhealthcare Teva Lonquex Referral Form for Amend Proof V2 - newworldhealthcare Fax Email Print - pdfFiller

Teva Pharmaceutical has been given a green light by the European Commission (EC) for Lonquex, a rival to Amgen's blockbuster Neulasta. | New Drug Approvals










![Buy Lonquex® [Lipegfilgrastim] Buy Lonquex® [Lipegfilgrastim]](https://pillbuys.com/26185-home_default/oncotron-mitoxantrone-.jpg)
