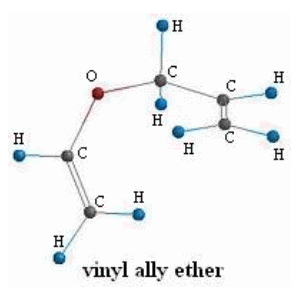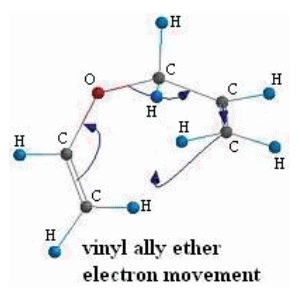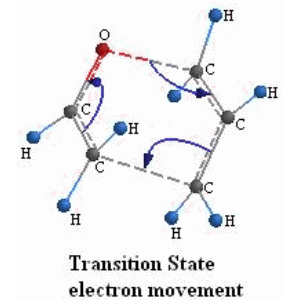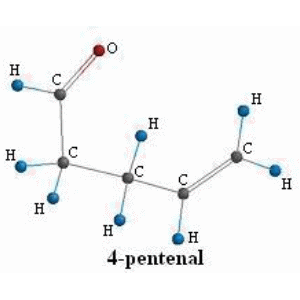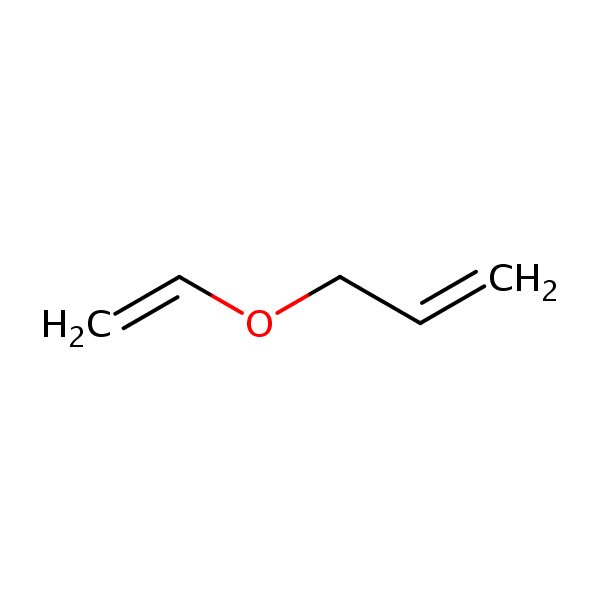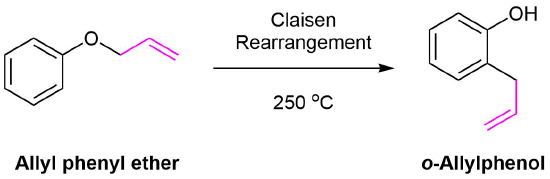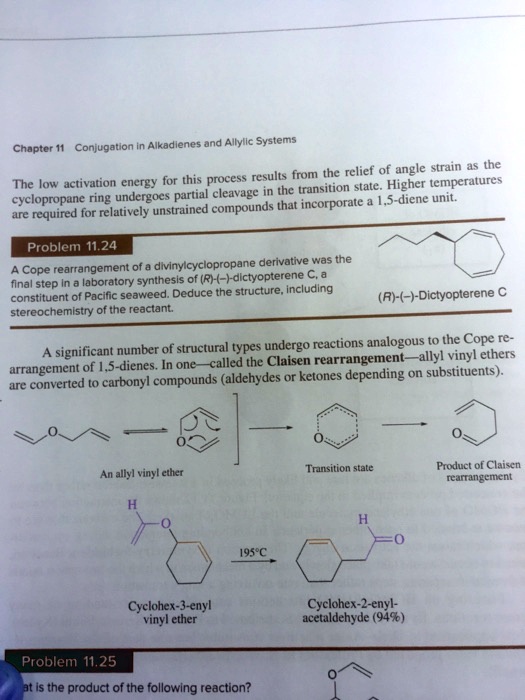
SOLVED:Conjugation Alkadlenes and _ Allyllc Systems Chapter 11 from the relief of angle strain as the The low activation energy for this process results transition state: Higher temperatures cyclopropane ring undergoes partial
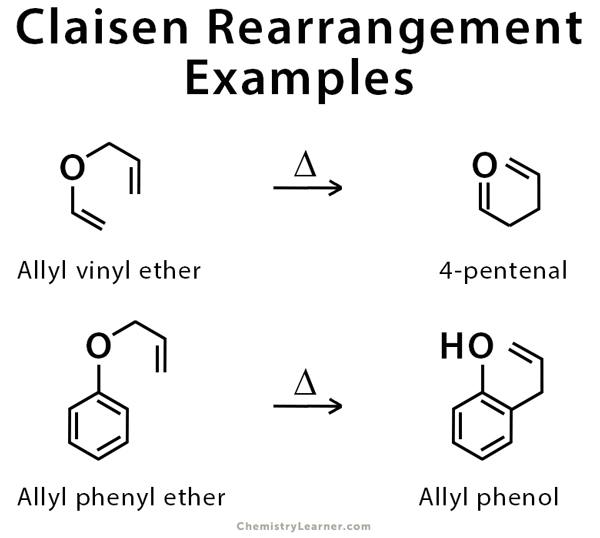
![PDF] Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9 | Semantic Scholar PDF] Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f7fadaa27890038feda7ad1d429351688b9cd998/5-Figure4-1.png)
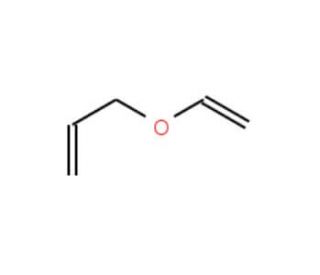
PDF] Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9 | Semantic Scholar
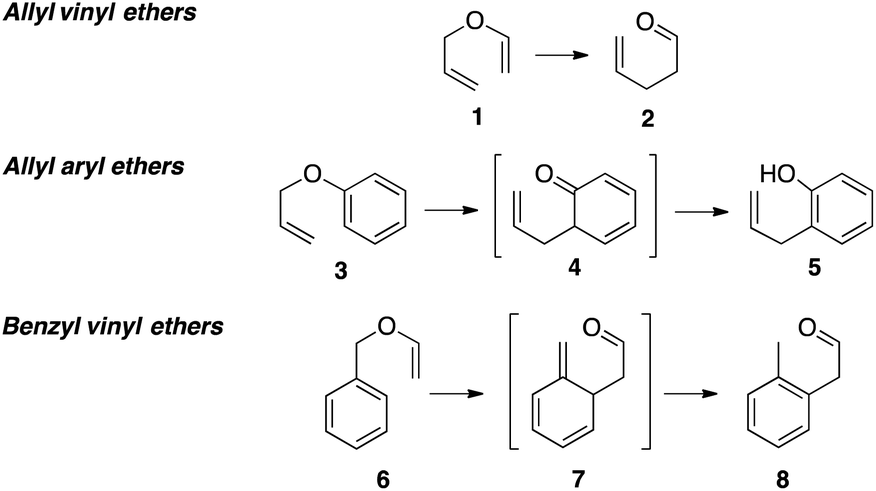
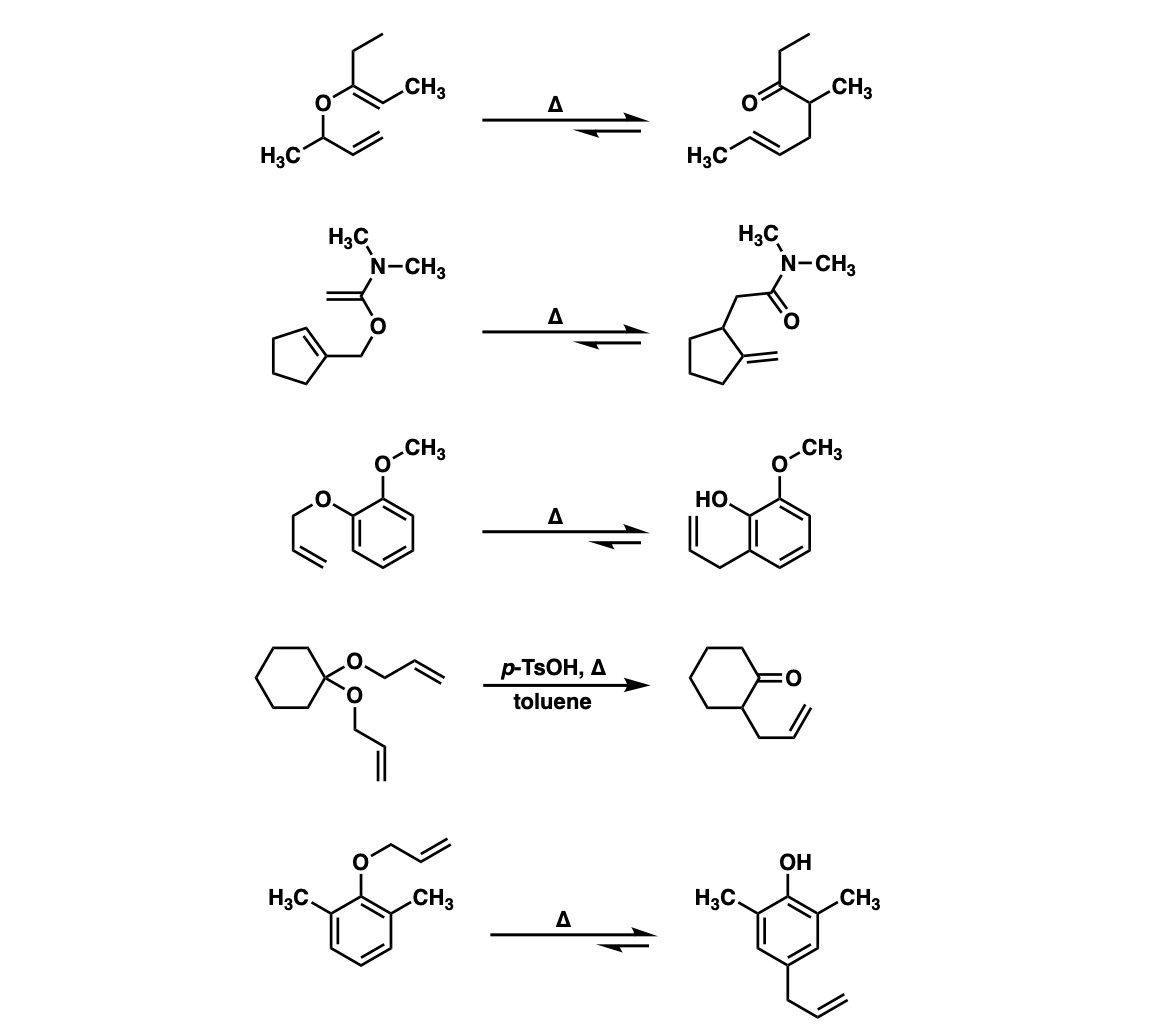
Claisen rearrangements of benzyl vinyl ethers: theoretical investigation of mechanism, substituent effects, and regioselectivity - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB01666B

Access to Vinyl Ethers and Ketones with Hypervalent Iodine Reagents as Oxy‐ Allyl Cation Synthetic Equivalents - Declas - 2020 - Angewandte Chemie International Edition - Wiley Online Library
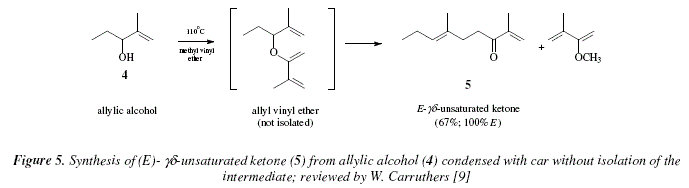
Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9
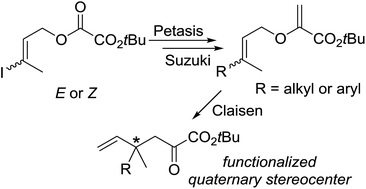
Modular synthesis of allyl vinyl ethers for the enantioselective construction of functionalized quaternary stereocenters - RSC Advances (RSC Publishing)

Predict the major products of the reaction: Allyl vinyl ether is treated with HI in equimolar ratio - Chemistry - - 10642779 | Meritnation.com

Activation of ether functionality of allyl vinyl ethers by chiral bis(organoaluminum) Lewis acids: application to asymmetric Claisen rearrangement - ScienceDirect

Allyl vinyl ethers via Wittig olefination: a short and efficient synthesis of (±)-mesembrine - ScienceDirect